Ihr medizintechnisches Team in Kiel

Aufgaben und Ziele unseres Unternehmens
Die Produkte, die wir entwickeln, herstellen und vertreiben, sollten den kranken und leidenden Menschen helfen, menschenwürdig zu leben.
Unsere Problemlösung orientiert sich an der Natur, um aus der Erfahrung der Evolution zu profitieren.
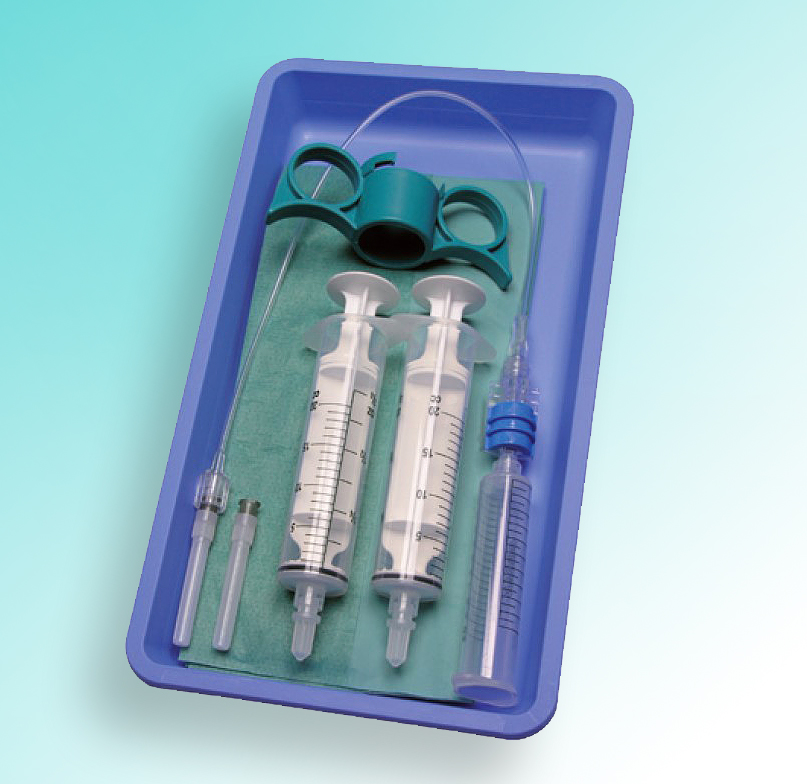
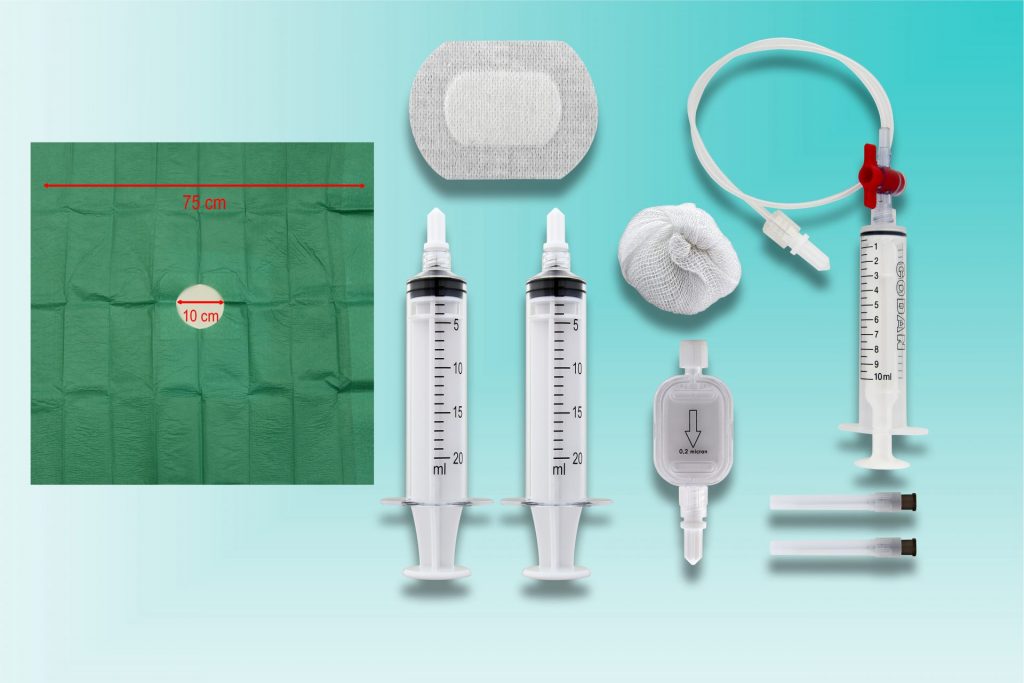
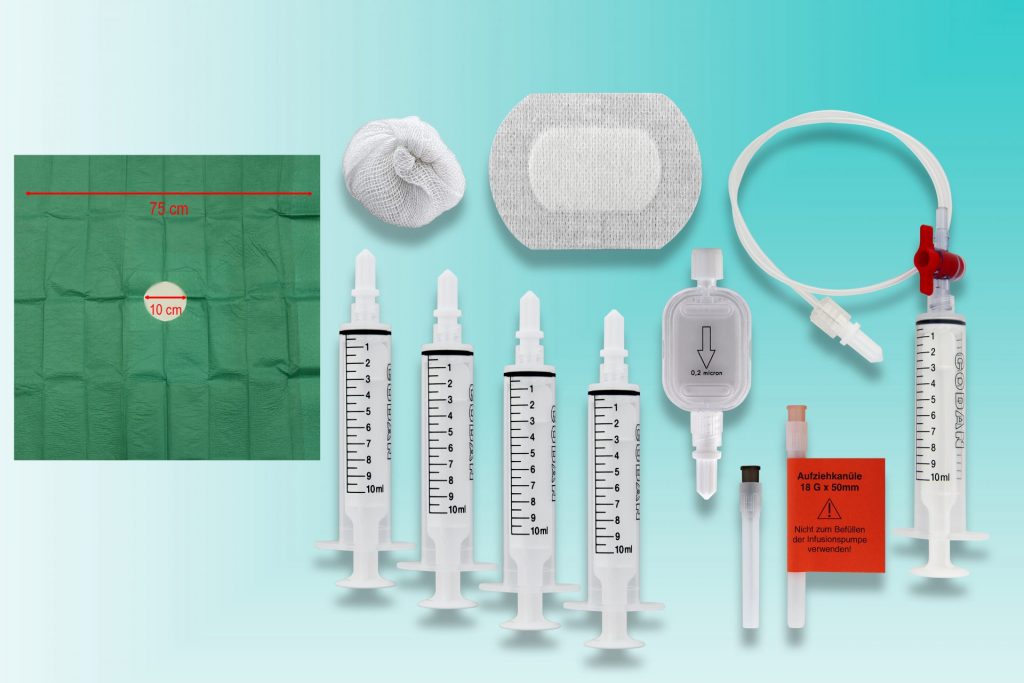
Unsere Produkte
*Hersteller Medizintechnik Promedt GmbH
**Hersteller Firma ECOMEDIS Medizintechnik GmbH
Kontaktieren Sie uns
Mit dem Laden der Karte akzeptieren Sie die Datenschutzerklärung von Google.
Mehr erfahren
PGlmcmFtZSBsb2FkaW5nPSJsYXp5IgoJCQkJCXNyYz0iaHR0cHM6Ly9tYXBzLmdvb2dsZS5jb20vbWFwcz9xPXRyaWN1bWVkJTIwJTIwUiVDMyVCNm50Z2Vuc3RyYSVDMyU5RmUlMjA3QSUyQyUyMDI0MTQzJTIwS2llbCYjMDM4O3Q9bSYjMDM4O3o9MTcmIzAzODtvdXRwdXQ9ZW1iZWQmIzAzODtpd2xvYz1uZWFyIgoJCQkJCXRpdGxlPSJ0cmljdW1lZCAgUsO2bnRnZW5zdHJhw59lIDdBLCAyNDE0MyBLaWVsIgoJCQkJCWFyaWEtbGFiZWw9InRyaWN1bWVkICBSw7ZudGdlbnN0cmHDn2UgN0EsIDI0MTQzIEtpZWwiCgkJCT48L2lmcmFtZT4=
geöffnet von 8:00 Uhr – 16:30 Uhr
Adresse Röntgenstraße 7A, 24143 Kiel